Training Service

Compliance Specialist
Global Regulatory Services
What We Offer
Empowering Compliance Through Expert
Training & Certification
Our professional training and certification programs are designed to equip teams with the regulatory knowledge and practical skills required to meet global compliance standards and drive quality excellence across medical device and life science organizations.
Professional Training Programs
Certified Experts
- Global Regulatory & Quality Training ISO 13485,EU MDR (2017/745) / IVDR (2017/746), US FDA QSR (Part 820), Risk Management (ISO 14971)
- Clinical & Compliance Excellence Programs Clinical Evaluation, Post-Market Surveillance, Regulatory Affairs & QA best practices
- Goal: Enable teams to maintain compliance, reduce regulatory risk, and achieve quality excellence.
We assess compliance needs and deliver customized, role-based training aligned with global regulations, ensuring audit readiness and regulatory confidence.
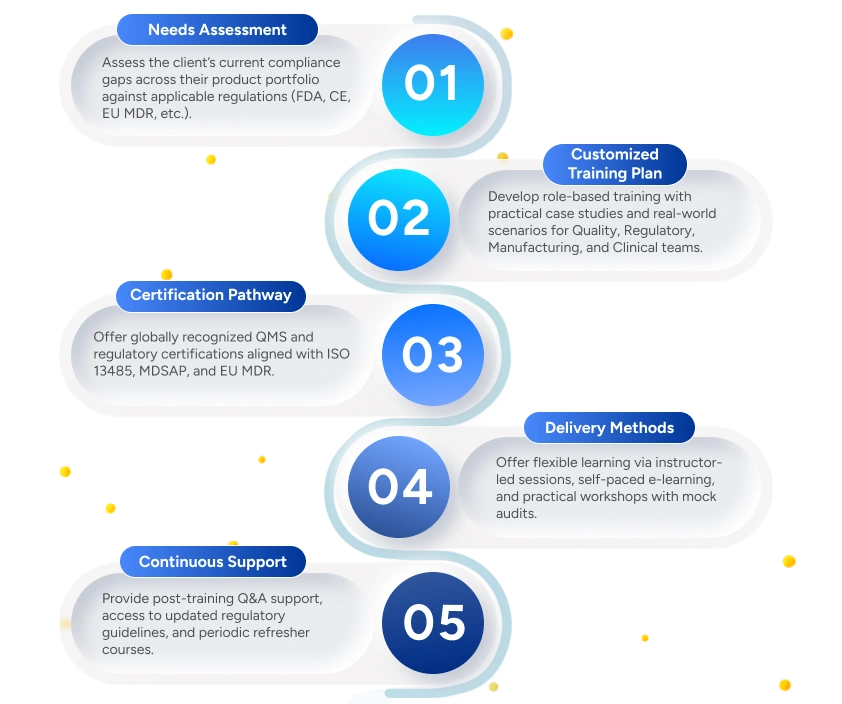
Deliverables to the Client

Deliverables
Training Materials
Includes role-based content, practical examples, and assessment tools to support effective learning.
Certification
Certificates serve as documented evidence of competence and regulatory compliance.
Compliance Toolkit
Supports practical implementation and ongoing compliance after training completion.
Gap Analysis Report
Identifies competency gaps and recommends targeted improvement actions.
Knowledge Portal Access
Enables continuous learning, refreshers, and access to the latest regulatory information.
Benefits
Regulatory Compliance Assurance
Reduces the risk of non-compliance, observations, and regulatory actions.
Improved Audit Readiness
Supports consistent documentation, responses, and audit performance.
Enhanced Quality Culture
Encourages proactive risk management and adherence to best practices.
Cost Savings
Reduces costs associated with audit failures, delays, and compliance issues.
Global Market Access
Facilitates smoother approvals and acceptance in multiple global markets.
Continuous Improvement
Drives ongoing optimization of processes, performance, and compliance.
Benefits for Your Organization

Call To Action
Simplify Compliance. Accelerate Market Access.
Our global team supports medical device and IVD manufacturers with end-to-end regulatory, quality, and clinical services-ensuring safe, effective, and compliant products at every stage.




.webp)