
Design Validation

Compliance Specialist
Global Regulatory Services
What We Offer
Design Verification & Validation Services
We provide design verification and validation services to ensure medical devices meet their intended use, performance expectations, and global regulatory requirements.
Design Validation
Regulatory Compliance
- Design validation & testing services, including biocompatibility compliance evaluation and ETO/Gamma sterilization validation.
- Product durability & performance studies, including shelf-life testing with accelerated aging and technical validation support.
- Post-market compliance support, including PMS plans and reports, PMCF plans and reports, and Product Safety Update Reports.
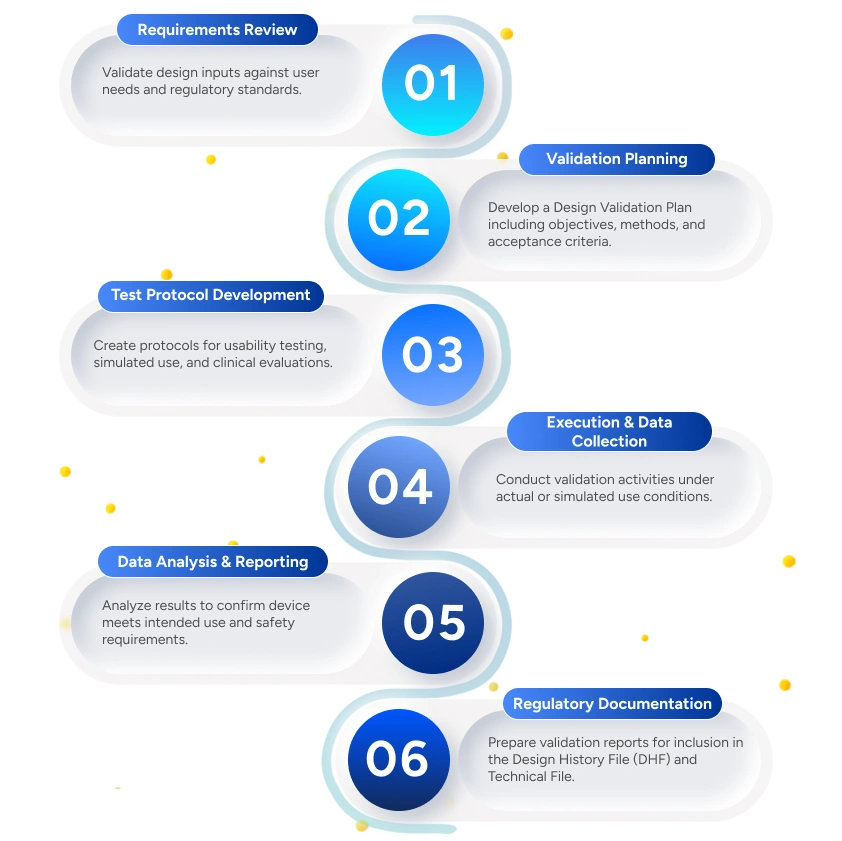
We ensure your medical device design meets user needs, intended use, and regulatory requirements through a structured Design Validation process aligned with ISO 13485 and EU MDR/FDA guidelines.
Deliverables to the Client

Deliverables
Design Validation Plan
Ensures design outputs meet user needs and intended use.
Validation Test Protocols
Aligned with applicable standards and regulatory expectations.
Usability & Simulated Use Test Reports
Preparation of reports documenting usability engineering and simulated-use testing outcomes.
Clinical Validation Reports (If Applicable)
Confirms clinical performance and safety for intended use.
Comprehensive Design Validation Report
Provides full traceability to design inputs, risks, and user requirements.
Regulatory Submission Support
Includes responses to regulatory queries and deficiency resolution support.
Benefits
Regulatory Compliance
Meet ISO 13485, EU MDR (2017/745) / IVDR (2017/746), and US FDA QSR Part 820 design validation requirements.
Risk Reduction
Ensure device safety and performance before market release.
User-Centric Design
Validate that the product meets real-world user needs.
Faster Approvals
Well-documented validation accelerates regulatory review.
Market Confidence
Demonstrates commitment to quality and patient safety.
Benefits for Your Organization

Call To Action
Simplify Compliance. Accelerate Market Access.
Our global team supports medical device and IVD manufacturers with end-to-end regulatory, quality, and clinical services-ensuring safe, effective, and compliant products at every stage.




.webp)