
Periodic Safety Update Report (PSUR)

Compliance Specialist
Global Regulatory Services
What We Offer
Periodic Safety Update Report (PSUR) Preparation & Maintenance
We prepare and maintain accurate PSURs to ensure continuous monitoring and reporting of medical device safety. Our approach ensures compliance with MDR/IVDR and supports proactive risk management.
PSUR Documentation
Safety Monitoring
- PSUR Preparation & Reporting Compilation of device safety data and trend analysis in compliance with MDR/IVDR
- Ongoing Safety & Compliance Support Periodic updates, risk assessment integration, and regulatory-ready documentation
We develop accurate EU MDR–aligned PSURs based on regulatory and device data analysis Structured documentation supports continuous safety reporting and compliance readiness
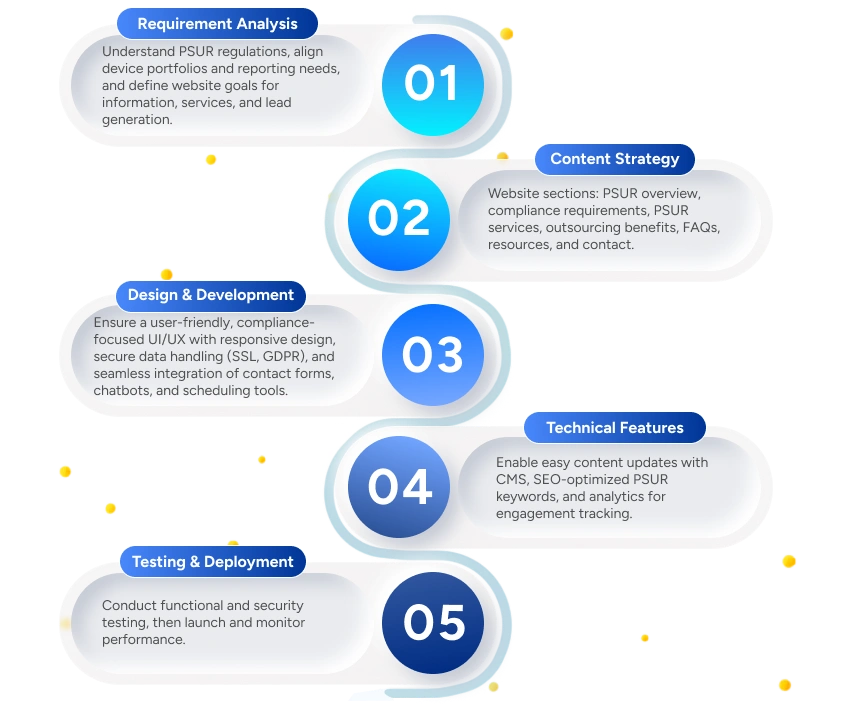
Deliverables to the Client

Deliverables
Fully Functional PSUR Service Website
Informative content on PSUR compliance.
Service description and process flow.
Contact and lead capture forms.
Resource library (guides, templates).
SEO-Optimized Pages for Better Visibility
Improves online visibility and search engine ranking for PSUR-related regulatory services.
Analytics Dashboard for Client Insights
Provides actionable insights to improve service outreach and client engagement.
Benefits
Regulatory Compliance Assurance
Supports organizations in understanding and meeting ongoing post-market obligations.
Brand Credibility
Builds confidence among manufacturers, notified bodies, and stakeholders.
Lead Generation
Converts website traffic into actionable business opportunities.
Educational Resource
Supports client awareness and informed decision-making on PSUR compliance.
Time & Cost Efficiency
Optimizes marketing and compliance outreach with minimal operational overhead.
Benefits for Your Organization

Call To Action
Simplify Compliance. Accelerate Market Access.
Our global team supports medical device and IVD manufacturers with end-to-end regulatory, quality, and clinical services-ensuring safe, effective, and compliant products at every stage.




.webp)