
Summary of Safety and Clinical Performance (SSCP)

Compliance Specialist
Global Regulatory Services
What We Offer
Summary of Safety and Clinical Performance (SSCP) Services
We prepare clear and compliant SSCP documents to communicate device safety and clinical performance to regulators and stakeholders. Our structured approach ensures transparency, regulatory compliance, and audit readiness.
SSCP Documentation
Clinical Transparency
- SSCP Preparation & Authoring Drafting and compiling safety and performance data in compliance with MDR/IVDR
- Regulatory & Stakeholder-Ready Reports Ensuring clear, accurate, and transparent communication to regulators and relevant parties
We assist medical device manufacturers in preparing SSCP documents that comply with EU MDR (2017/745) requirements, ensuring transparency and regulatory acceptance.
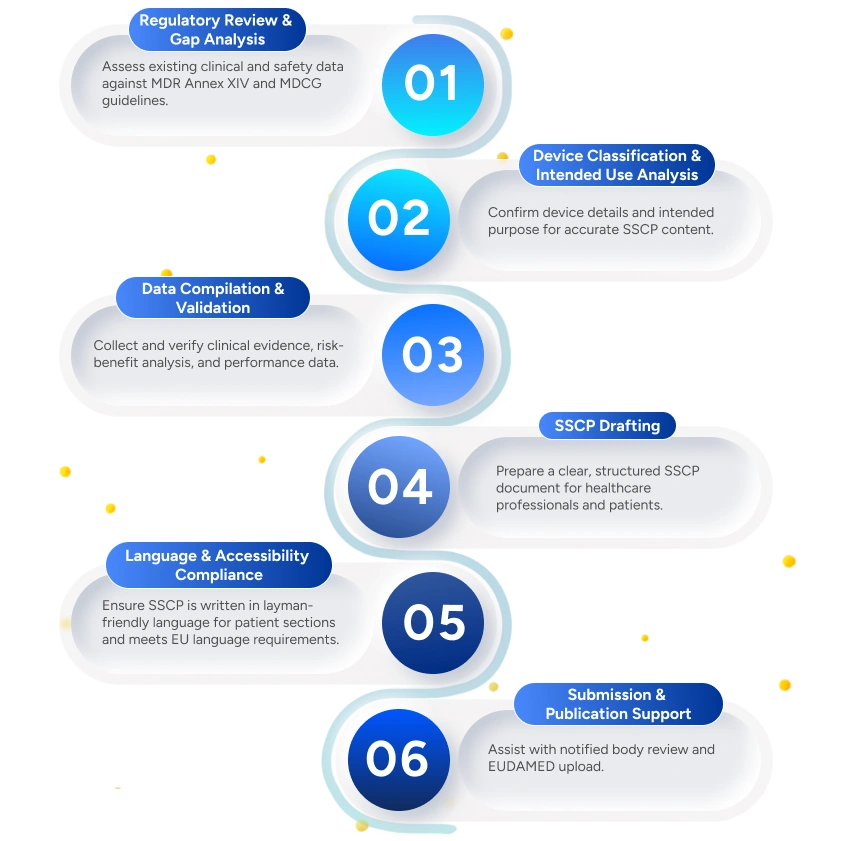
Deliverables to the Client

Deliverables
Gap Analysis Report
Assessment of existing clinical, safety, and PMS data against SSCP requirements under EU MDR.
Validated Clinical & Safety Data Summary
Ensures consistency, accuracy, and regulatory traceability.
Draft SSCP Document
Healthcare Professional & Patient Sections
Risk-Benefit Analysis Summary
Summarized evaluation of clinical benefits versus known and residual risks.
EUDAMED Upload Support
Support for formatting, validation, and uploading SSCP documents to EUDAMED.
Update Strategy for Lifecycle Maintenance
Definition of triggers, frequency, and responsibilities for SSCP updates.
Benefits
Regulatory Compliance
Meet EU MDR SSCP requirements for CE marking.
Transparency & Trust
Provide clear safety and performance information to users and professionals.
Market Access
Smooth notified body review and faster approvals.
Risk Management
Demonstrate benefit-risk ratio with robust evidence.
Lifecycle Support
Keep SSCP updated with PMS and PMCF data.
Benefits for Your Organization

Call To Action
Simplify Compliance. Accelerate Market Access.
Our global team supports medical device and IVD manufacturers with end-to-end regulatory, quality, and clinical services-ensuring safe, effective, and compliant products at every stage.




.webp)