
QMS Documentation and Implementation

Compliance Specialist
Global Regulatory Services
What We Offer
QMS Documentation & Regulatory-Compliant Implementation
We design, document, and implement robust Quality Management Systems that align with global regulatory standards, helping organizations achieve compliance, audit readiness, and sustainable quality operations.
QMS Documentation & Implementation
QMS Implementation
- Developing comprehensive QMS documentation—including Quality Manual, procedures, SOPs, work instructions, and formats-aligned with ISO 13485, US FDA 21 CFR Part 820 (QMSR), EU MDR/IVDR, IMDR, and MDSAP requirements.
- Implementing an integrated, risk-based QMS ensuring regulatory compliance, design controls, DHF/DMR/DHR, 510(k), traceability, and continual improvement.
- Establishing an audit-ready system covering technical documentation, risk management, clinical evaluation, UDI, PMS, vigilance, and complete global inspection preparedness.
We help medical device organizations achieve ISO 13485 compliance through a structured and efficient process
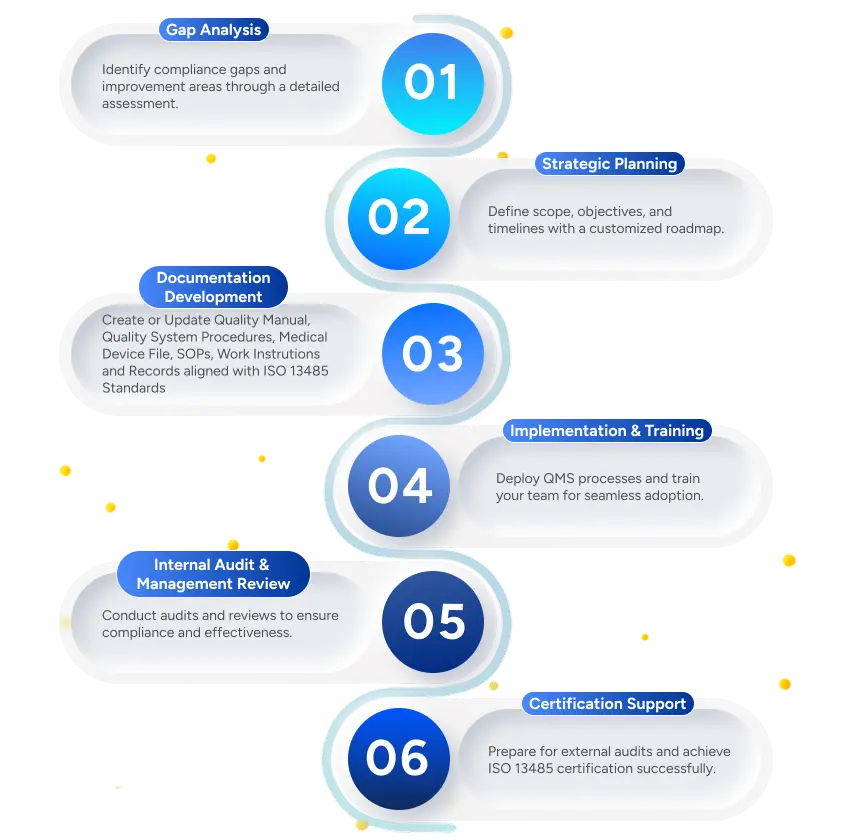
Deliverables to the Client

Deliverables
Gap Analysis Report
Comprehensive Gap Analysis Report
QMS Documentation
Customized QMS Documentation (Policies, SOPs, Work Instructions)
Training Materials & Sessions
To ensure awareness, competence, and effective QMS implementation across the organization.
Internal Audit Reports
To verify QMS conformity and effectiveness prior to certification.
Management Review Templates
To support top management in evaluating QMS performance and strategic alignment.
Certification Readiness Checklist
To confirm full preparedness for external certification audit.
Benefits
Regulatory Compliance
Meet global medical device standards.
Risk Reduction
Minimize product and process risks.
Operational Efficiency
Streamlined processes reduce errors and costs.
Global Market Access
ISO 13485 certification opens international opportunities.
Customer Confidence
Demonstrates commitment to quality and safety.
Continuous Improvement
Built-in mechanisms for ongoing enhancement
Benefits for Your Organization

Call To Action
Simplify Compliance. Accelerate Market Access.
Our global team supports medical device and IVD manufacturers with end-to-end regulatory, quality, and clinical services-ensuring safe, effective, and compliant products at every stage.




.webp)