
Clinical Investigation Report (CIR)

Compliance Specialist
Global Regulatory Services
What We Offer
Clinical Investigation Report (CIR) Preparation & Documentation
We prepare comprehensive Clinical Investigation Reports to document clinical studies in compliance with GCP and regulatory requirements. Our structured approach supports approvals, audit readiness, and lifecycle compliance.
CIR Documentation
Clinical Compliance
- CIR Preparation & Authoring Complete documentation of clinical investigations, study protocols, and results in compliance with GCP.
- Regulatory-Ready Reporting Ensures CIRs meet global regulatory standards and are audit-ready.
We help medical device manufacturers prepare Clinical Investigation Reports (CIR) that comply with EU MDR (2017/745) and ISO 14155 requirements, ensuring robust documentation of clinical investigations.
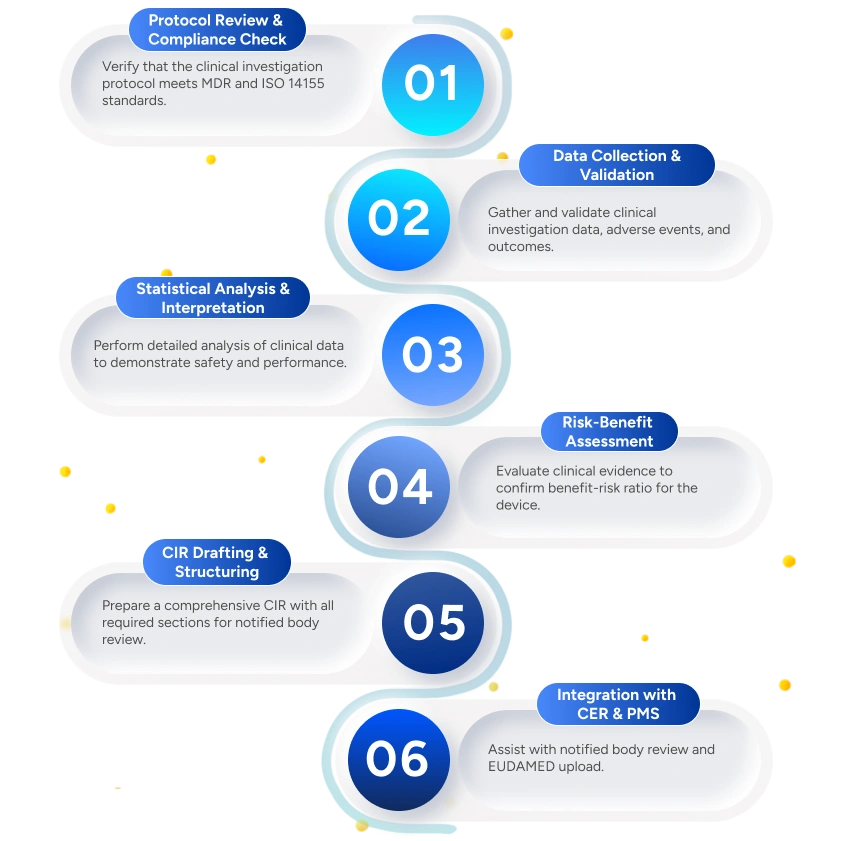
Deliverables to the Client

Deliverables
Clinical Investigation Protocol Review Summary
Summarizes protocol alignment, deviations, and key implementation considerations.
Validated Clinical Data Tables & Analysis
Preparation and validation of clinical data tables, listings, and statistical analyses.
Risk-Benefit Assessment Report
Evaluation of clinical outcomes against identified and residual risks.
Comprehensive Clinical Investigation Report (CIR)
Documents study conduct, results, deviations, and conclusions.
Integration Plan with CER & PMS
Ensures consistency across clinical evidence and lifecycle documentation.
Regulatory Submission Support
Includes response management for authority queries and follow-up requests.
Benefits
Regulatory Compliance
Meet EU MDR and ISO 14155 requirements for clinical investigations.
Market Access
Smooth notified body review and faster CE marking approvals.
Risk Mitigation
Identify and address safety concerns proactively.
Evidence-Based Assurance
Demonstrate device safety and performance with robust clinical data.
Lifecycle Support
Keep CIR aligned with CER and PMS for continued compliance.
Benefits for Your Organization

Call To Action
Simplify Compliance. Accelerate Market Access.
Our global team supports medical device and IVD manufacturers with end-to-end regulatory, quality, and clinical services-ensuring safe, effective, and compliant products at every stage.




.webp)