
Product Registration Services

Compliance Specialist
Global Regulatory Services
What We Offer
End-to-End Medical Device Product
Registration Services
End-to-end regulatory affairs and market access support for medical device companies, covering US FDA 510(k) and Pre-Submission applications, Europe Technical Files and CE submissions, and country filings including Health Canada, Israel (AMAR), China (CFDA), Japan (MHLW), Korea (KFDA), and Taiwan (TFDA), along with notified body selection, FDA establishment registration, and alignment with IMDR and MDSAP for global approvals.
Regulatory Submissions
Global Market Access
- Global regulatory submissions: US FDA 510(k), Pre-Sub, CE Technical Files, Health Canada, AMAR, CFDA, MHLW, KFDA, and TFDA filings.
- Regulatory affairs & market access: Strategy development, notified body selection, and US FDA establishment registration for manufacturers.
- Comprehensive compliance support: Assistance for CE marking, FDA submissions, IMDR, MDSAP, and global market approvals.
We simplify the complex process of medical device product registration by ensuring compliance with global regulatory requirements.
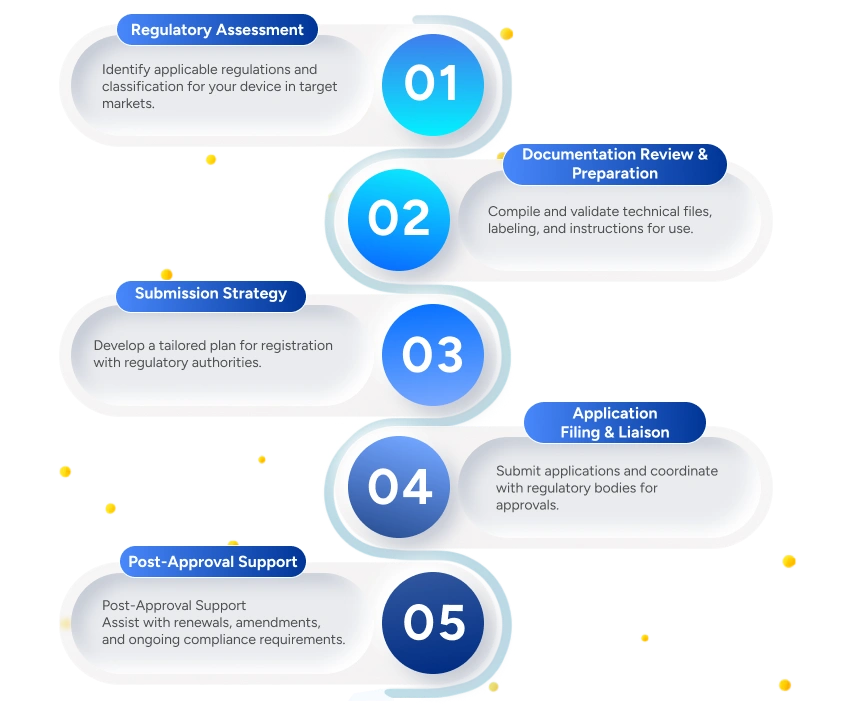
Deliverables to the Client

Deliverables
Regulatory Pathway Report
Identifies the applicable regulatory framework, approval route, and registration strategy for the medical device in the target market.
Device Classification & Compliance Checklist
Determines the medical device classification based on intended use and risk level.
Complete Technical Documentation
Ensures alignment with regulatory authority expectations and submission requirements.
Registration Application Submission
Includes coordination for queries, deficiency responses, and regulatory follow-ups.
Approval Confirmation & Certificates
Verification of approval scope and guidance on usage of certificates and approvals.
Post-Market Compliance Guidance
Support for change management, renewals, and ongoing regulatory compliance.
Benefits
Faster Market Entry
Streamlined registration process reduces delays.
Global Compliance
Meet regulatory requirements in multiple jurisdictions.
Risk Mitigation
Avoid costly errors and non-compliance penalties.
Expert Guidance
Leverage our experience for smooth approvals.
Customer Trust
Certified products enhance credibility and market acceptance.
Benefits for Your Organization

Call To Action
Simplify Compliance. Accelerate Market Access.
Our global team supports medical device and IVD manufacturers with end-to-end regulatory, quality, and clinical services-ensuring safe, effective, and compliant products at every stage.




.webp)