
Biostatistics & Medical Writing

Compliance Specialist
Global Regulatory Services
What We Offer
Biostatistics & Medical Writing Services
We provide expert statistical analysis and medical writing support for clinical and regulatory submissions. Our services ensure accurate, compliant, and high-quality documentation for approvals and audits.
Biostatistical Analysis
Regulatory Writing
- Statistical Analysis & Data Support Study design, data evaluation, and interpretation for clinical and regulatory submissions
- Medical & Regulatory Writing Preparation of reports, manuscripts, and regulatory documents in compliance with global standards
We provide expert biostatistical analysis and medical writing solutions to support clinical research, regulatory submissions, and scientific publications. Our approach ensures accuracy, compliance, and clarity.
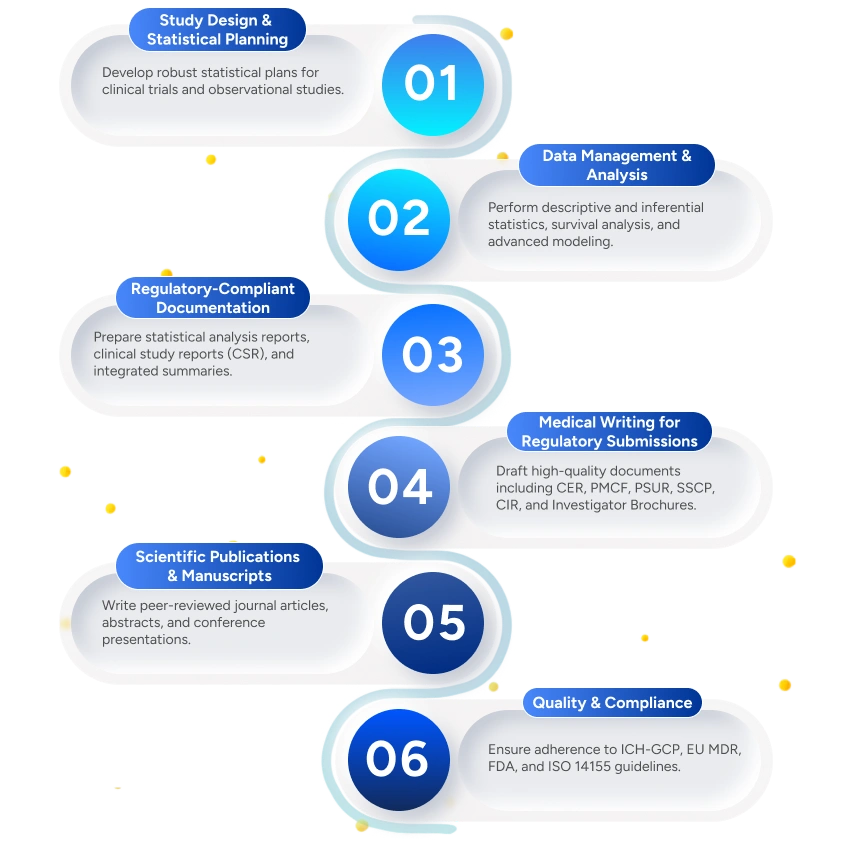
Deliverables to the Client

Deliverables
Statistical Analysis Plan (SAP)
Pre-specified to ensure unbiased and reproducible analysis.
Validated Statistical Reports
Preparation of verified statistical outputs, tables, listings, and figures.
Clinical Study Reports (CSR)
Summarizes study design, conduct, analysis, and clinical conclusions.
Regulatory Documents (CER, PMCF, PSUR, SSCP, CIR)
Ensures consistency, scientific rigor, and compliance across submissions.
Manuscripts & Publications
Includes journal targeting, formatting, and response to reviewer comments.
Data Visualization & Interpretation
Supports meaningful interpretation of clinical and statistical results.
Benefits
Regulatory Compliance
Meet global standards for clinical and regulatory documentation.
Data Integrity
Accurate statistical analysis ensures reliable conclusions.
Faster Approvals
Well-structured reports streamline regulatory review.
Scientific Credibility
High-quality publications enhance reputation.
End-to-End Support
From study design to submission-ready documents.
Benefits for Your Organization

Call To Action
Simplify Compliance. Accelerate Market Access.
Our global team supports medical device and IVD manufacturers with end-to-end regulatory, quality, and clinical services-ensuring safe, effective, and compliant products at every stage.




.webp)