
Regulatory


Compliance Specialist
Global Regulatory Services
What We Offer
Comprehensive Regulatory Support Services
We provide end-to-end regulatory assistance to help medical device manufacturers obtain approvals and maintain compliance across global markets, including CDSCO, CE, US FDA QSR Part 820, and other international requirements.
Regulatory Assistance
Global Approvals
- Global Regulatory Submissions CDSCO licenses, CE marking, US FDA QSR Part 820 submissions, and international approvals
- Compliance & Lifecycle Support IMDR, MDSAP, and ongoing regulatory compliance management
CE Marking (EU MDR / IVDR / SaMD)


SaMD
CE MDR
We provide comprehensive Regulatory Affairs (RA) services to help medical device manufacturers achieve global compliance and market access.
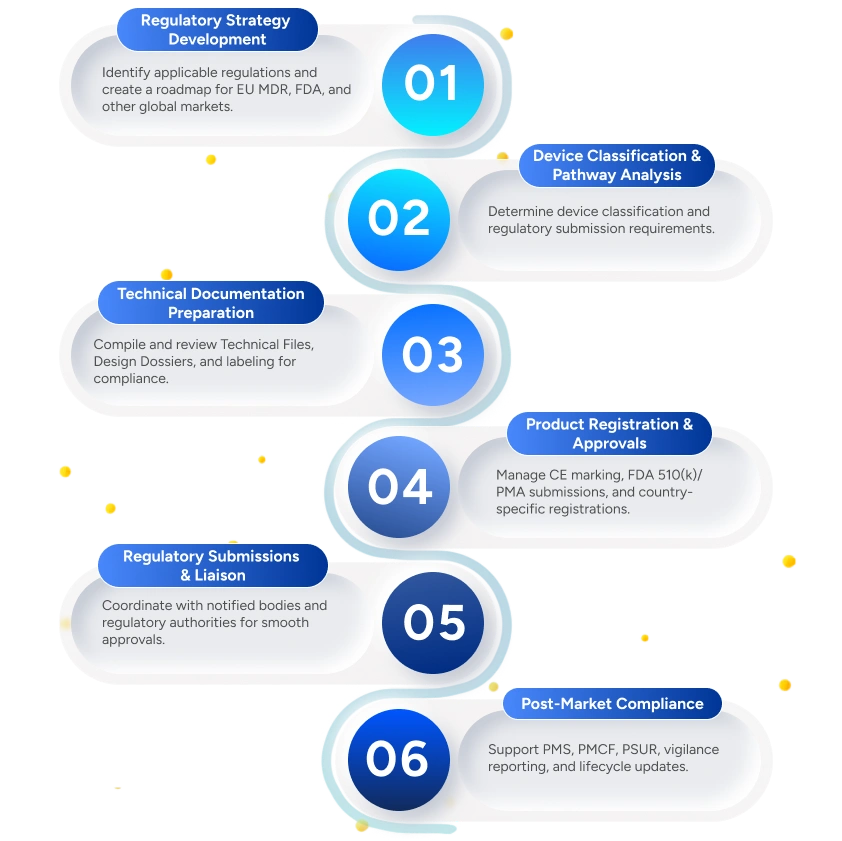
Deliverables to the Client

Deliverables
Regulatory Compliance Roadmap
Defines the regulatory strategy, applicable regulations, and approval pathways for target markets.
Device Classification Report
Determines device classification based on intended use and risk profile.
Complete Technical Documentation
Preparation of comprehensive technical files/design dossiers covering design, risk, clinical, and manufacturing data.
Product Registration Certificates
Support through regulatory review until official approvals and registration certificates are obtained. Includes confirmation of approved scope and registration details.
Regulatory Submission Packages
Compilation and validation of complete submission dossiers for regulatory authorities.
Post-Market Surveillance & Vigilance Reports
Preparation of PMS plans, PSUR/PMS reports, and vigilance documentation.
Benefits
Global Market Access
Achieve compliance for EU, US, and other regions.
Risk Mitigation
Avoid costly delays and non-compliance penalties.
Faster Approvals
Streamlined submissions and proactive regulatory planning.
Quality Assurance
Ensure documentation meets notified body expectations.
Continuous Support
Stay updated with evolving regulations and standards.
Benefits for Your Organization

Call To Action
Simplify Compliance. Accelerate Market Access.
Our global team supports medical device and IVD manufacturers with end-to-end regulatory, quality, and clinical services-ensuring safe, effective, and compliant products at every stage.




.webp)