
QA/RA Advisory

Compliance Specialist
Global Regulatory Services
What We Offer
Strategic QA/RA Advisory for Regulatory
Compliance & Approvals
Our QA/RA advisory services provide expert guidance to navigate global regulatory requirements and quality standards. We help organizations streamline compliance activities, reduce regulatory risks, and accelerate approvals. Our strategic support ensures sustained compliance throughout the product lifecycle.
QA/RA Advisory
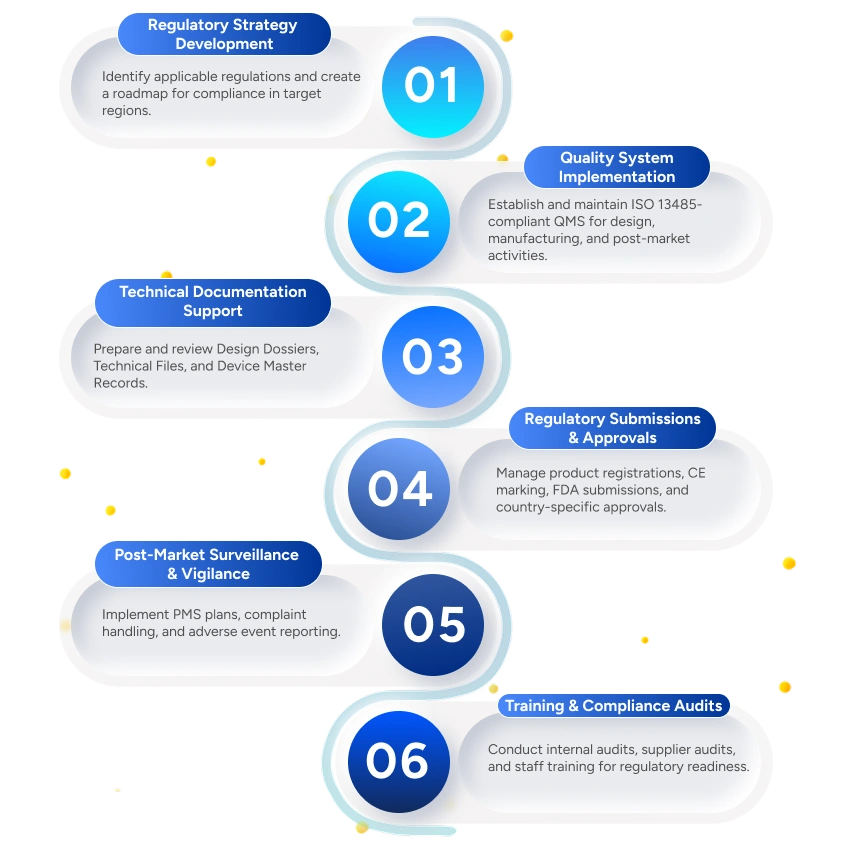
Regulatory Strategy
- QA/RA advisory services to streamline regulatory compliance, accelerate market approvals, and align quality systems with global requirements.
- Clinical evidence development and reporting, including Clinical Evaluation Report (CER), Post Market Clinical Follow-up (PMCF), Summary of Safety and Clinical Performance (SSCP), and Clinical Investigation Report (CIR).
- Post-market safety and analytics support, including Periodic Safety Update Report (PSUR), biostatistics, and medical writing for regulatory submissions.
We provide end-to-end Quality Assurance (QA) and Regulatory Affairs (RA) solutions to help medical device manufacturers achieve compliance, maintain quality, and access global markets.
Deliverables to the Client

Deliverables
Regulatory Compliance Roadmap
Defines the regulatory strategy, applicable standards, and approval pathways for target markets.
ISO 13485 QMS Documentation
Development and customization of ISO 13485–compliant policies, procedures, and records.
Technical File / Design Dossier Preparation
Compilation of complete technical documentation covering design, manufacturing, risk, and clinical evidence.
Product Registration & Approval Certificates
Support for regulatory submissions and coordination with authorities until approval is obtained.
Post-Market Surveillance Reports
Preparation of PMS plans, PSUR/PMS reports, and vigilance documentation.
Audit Reports & CAPA Plans
Conduct of internal and supplier audits with documented findings and compliance status.
Training Materials & Regulatory Updates
Development of role-based training materials for QA/RA and operational teams.
Benefits
Global Market Access
Meet regulatory requirements for EU, US, and other regions.
Risk Reduction
Avoid compliance gaps and costly penalties.
Faster Approvals
Streamlined submissions and proactive regulatory planning.
Quality Excellence
Robust QMS ensures product safety and reliability.
Customer Confidence
Demonstrates commitment to quality and compliance.
Continuous Support
Stay updated with evolving regulations and standards.
Benefits for Your Organization

Call To Action
Simplify Compliance. Accelerate Market Access.
Our global team supports medical device and IVD manufacturers with end-to-end regulatory, quality, and clinical services-ensuring safe, effective, and compliant products at every stage.




.webp)